
Sample size re-estimation for superiority clinical trials with a dichotomous outcome using an unblinded estimate of the control group outcome rate

Sample size re-estimation without un-blinding for time-to-event outcomes in oncology clinical trials
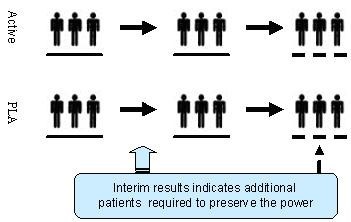
Adaptive design methods in clinical trials – a review | Orphanet Journal of Rare Diseases | Full Text
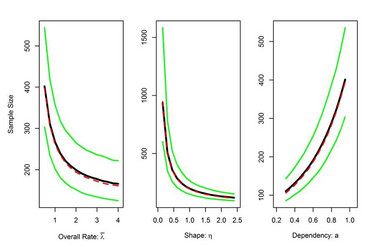
Blinded Sample Size Reestimation in Clinical Trials with Recurrent Event Data and Time-dependent Event Rates

Design for sample size re-estimation with interim data for double-blind clinical trials with binary outcomes. | Semantic Scholar

Sample size calculation and re-estimation based on the prevalence in a single-arm confirmatory diagnostic accuracy study - Maria Stark, Antonia Zapf, 2020

Adaptive Features of a Trial That Uses Sample-Size Reestimation. The... | Download Scientific Diagram
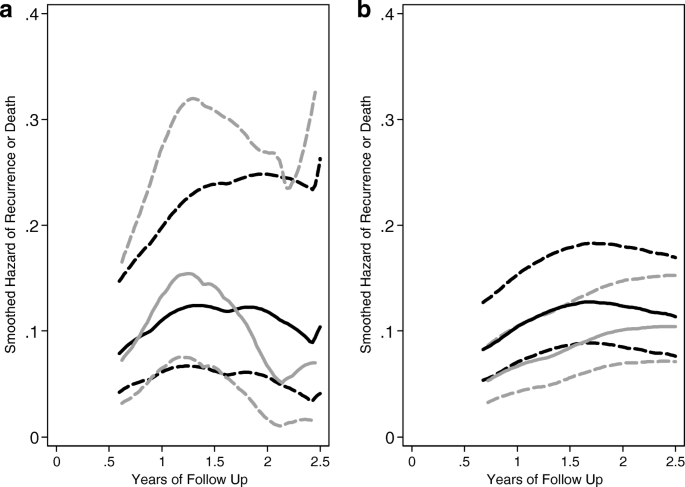
Follow up after sample size re-estimation in a breast cancer randomized trial for disease-free survival | Trials | Full Text

PPT - Adaptive Designs Sample size re-estimation: A review and recommendations PowerPoint Presentation - ID:1270910
Sample size re-estimation without unblinding for normally distributed outcomes with unknown variance: Communications in Statistics - Theory and Methods: Vol 21, No 10










